100 years of history
Throughout 100 years, we overcame challenges, celebrated victories, and built a legacy in health and science, with patients and life in mind. Discover some important moments of BIAL’s history on our timeline.
We remain inspired by a common vision of globally improving health and well-being.
We remain inspired by a common vision of globally improving health and well-being.


1924
Álvaro Portela founds BIAL
In 1924, Álvaro Portela (1894-1961) founded a medicine production industry in Porto, and he called it BIAL. It is now an innovative company on an international scale.

1929
The first BIAL product: Benzo-Diacol
The company’s first and very successful product trademark product was registered in 1929. This was Benzo-Diacol cough syrup and Álvaro Portela used a rather creative marketing campaign for it. One of the examples was putting advertising posters in some churches – particularly in Fátima – with the slogan “for silence in the chapel, Benzo-Diacol”.

1935
A new location for BIAL
BIAL moves to Rua João Oliveira Ramos, in Porto, where it remained until 1996.

1962
António Emílio Portela takes over the running of BIAL
In the 1960s, BIAL was already one of the largest Portuguese pharmaceutical companies, as a result of a focusing strongly on mechanization – under the leadership of Álvaro Portela’s son, António Emílio Portela.
1978
A new product arrives: Reumon is launched
BIAL launches Reumon, a topical anti-inflammatory gel with great success on the domestic market.

1979
Luís Portela takes over the running of BIAL (Chairman and CEO)
In early 1979, Luís Portela, a medical graduate, bought the majority of the capital in the company and became chairman of the board at the age of only 27. Three main strategic development lines were defined: quality, innovation and internationalization.

1984
First BIAL Award
Set up with the goal of encouraging medical research and disseminating work that had a significant impact on the area of medical research, the BIAL Award was created. From 1994, the Award was managed by the BIAL Foundation.

1992
The first R&D department
In late 1992, BIAL set up its Research and Development Department under the guidance of pharmacologist José Garrett, who was later succeeded by another pharmacologist, Patrício Soares da Silva.

1994
Setup of the BIAL Foundation
Committed to encouraging scientific studies on the Human Being – physical and spiritual – the BIAL foundation was set up in 1994. Led by Luís Portela, it is a benchmark institution, particularly in the area of research into Neuroscience and Parapsychology, aimed at stimulating discoveries that benefit people and make them healthier and reaching new heights in knowledge.

1996
Opening of the facilities in São Mamede do Coronado
It was in 1996 that BIAL left the center of Porto and the BIAL facilities in São Mamedo do Coronado were opened – a pivotal point in the growth of the company.
The first steps towards internationalisation
After the acquisition of a company named Aristegui, headquartered in Madrid and with a factory in Bilbao, BIAL founded its first affiliate in Spain. That same year also saw the set-up of Medimport in Mozambique – laying the first international foundations of BIAL. This year, the company also obtains the ISO 9001 Quality Standard.

2001
BIAL obtains ISO 14001
Based on the principle of excellence in all its activities, BIAL obtains ISO 14001 Environmental Management Systems Standard.

2005
Integration into EFPIA
In 2005, BIAL became a full member of the European Federation of Pharmaceutical Industries and Associations (EFPIA), an entity whose members are leading research companies in the European pharmaceutical sector.

2008
The increasing globalization of BIAL
2008 was an important year in terms of the company’s globalisation, with the opening of affiliates in two continents: Africa (Ivory Coast and Angola) and Central America (Panama).
2009
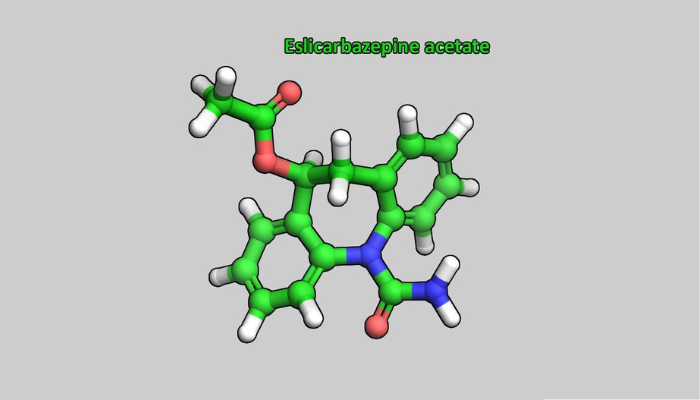
Launch of Zebinix®
The launch of Zebinix® (eslicarbazepine acetate), BIAL’s first research drug and the first patented Portuguese drug – for the treatment of epilepsy – left an indelible mark on the year 2009 and the history of BIAL. This antiepileptic drug was a result of over 15 years of the company focusing on research and development.

2010
A new affiliate of BIAL, this time in Italy.
It was in 2010 that BIAL took over the S.A.R.M Allergeni pharmaceutical company – specializing in the area of allergen immunotherapy – thus establishing its presence in Italy.

Luís Portela becomes Non-Executive Chairman and António Portela CEO
With BIAL having a strategic project based on Research and Development, as well as internationalization, Luís Portela decided to prepare his exit in a timely manner. After an internal discussion process, António Portela assumes the executive presidency of BIAL.

2012
Opening of a new vaccine factory in Bilbao
Another important moment was the opening in 2012 of the new vaccine factory in the Bilbao Technology Park, which has a Research and Development Department in the area of allergology. Later, in 2017, BIAL was to sell the allergology and immunology area to focus on the development of innovative drugs in its facilities in Portugal.

2013
FDA approves Aptiom®
Aptiom® (eslicarbazepine acetate) is approved by the Food and Drug Administration (FDA), thus beginning the commercialisation of a Portuguese medicine in the USA.

2015
BIAL begins operating in Germany and the United Kingdom
Following the strong focus on international expansion, BIAL opened its own facilities in Frankfurt and London.

2016
Launch of Ongentys®
Ongentys® (opicapone), the second drug from the company’s own research, is launched after approval by the European Commission.
Developed for the treatment of Parkinson’s, it is now a global drug present in the lives of around 80,000 patients all over the world.
Developed for the treatment of Parkinson’s, it is now a global drug present in the lives of around 80,000 patients all over the world.
2020
Ongentys® approval and new affiliates
BIAL announces that the US FDA (Food and Drug Administration) approved Ongentys® (opicapone). Japanese, Taiwanese and Australian regulatory authorities also approve Ongentys®.
Internationalisation remains key for BIAL and as such a new European affiliate was created, this time in Switzerland, and in the US, BIAL Biotech is also created.

2021
António Horta-Osório becomes the new Chairman of BIAL
António Horta Osório becomes Chairman of the Board of Directors of BIAL, replacing Luís Portela, the company's majority shareholder and chairman for 42 years. Luís Portela is still the Chairman of the BIAL Foundation.

2022
Opening of a new antibiotics factory and extension of the BIAL Campus
In October 2022, a new antibiotics production unit was opened, and the BIAL group’s industrial building was extended – as a result of the BIAL Campus expansion program. This meant a reinforcement of its production capacity and autonomy in the supply of drugs to the different markets where the company operates.

2023
R&D Strategic expansion
BIAL decides a strategic expansion of its Research and Development (R&D) area, starting its research into New Modalities and Rare Diseases.
Test your knowledge about the history of BIAL in a quiz with 10 questions.
Take part in our quiz!

